
Fluorine is a highly reactive element, and form fluorides with the majority of other. 
As you go up a group the amount of shells an atom has decreases which pulls the valence electrons in the outermost shell closer to the nucleus and increases an atom’s ability to attract more electrons To The protons in its nucleus. Chemistry for the gifted and talented: trends in reactivity in the periodic table Life cycle assessment: shopping bags 1416 years. In the second and final part of this series, we look deeper into reactivity trends in the periodic table, specifically focusing on halogens. Francium is the most reactive metal! reactivity of non-metals: As you go left to right within a period, the atom is getting a closer to obtaining a full valence shell, so the more readily it will react to receive those electrons. As you go down a group, the valence electrons move farther away from the nucleus (more shells) which make them easier to be given/taken away. As the electrons move closer to the nucleus, It will require more energy to remove them (High ionization energy) and It will be easier for the atom to attract more electrons (High electronegativity).Ĥ REACTIVITY OF Metals: As you go from right to left within a period, the amount of valence electrons a Metal has to give away decreases, so It will become more readily available to react. 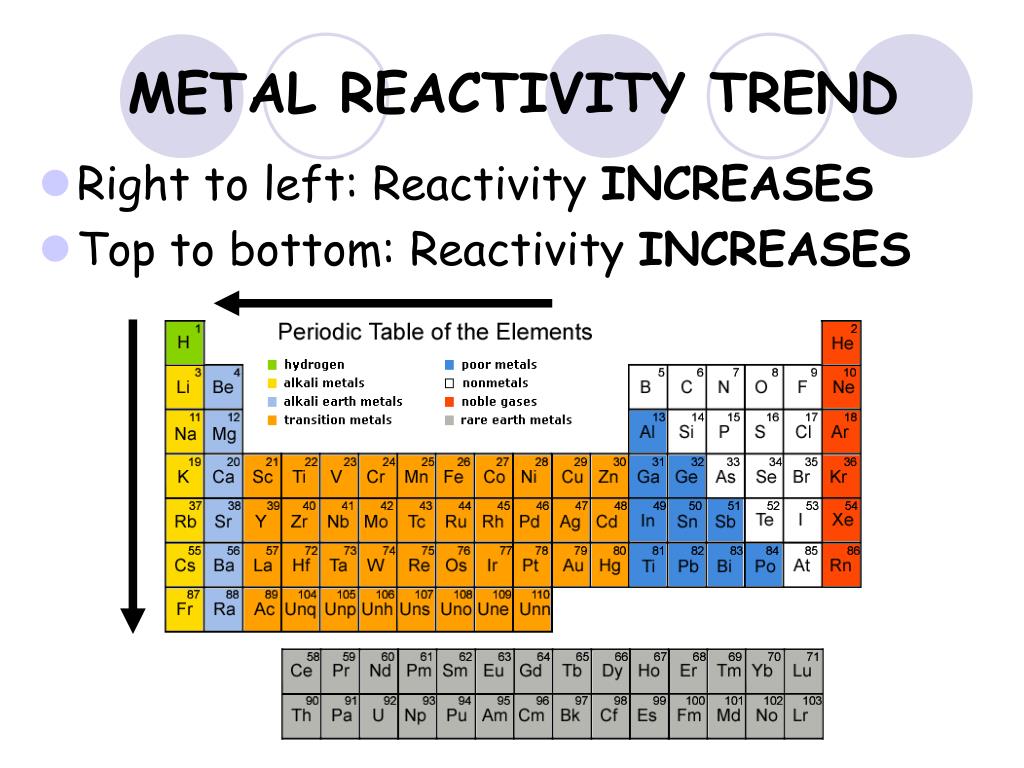
As the number of Protons and valence electrons Increase, The attraction increases which pulls the outermost shell closer to the nucleus (Opposites Attract). 2.6.6 summarizes the periodic trend in the properties of the elements. The atomic size and the metallic character are opposite, i.e., they increase from right to left in a row and from top to bottom in a column. Thus, it will require less energy (lower Ionization energy) to remove an electron from the outermost shell and It will also be more difficult To attract electrons (low electronegativity)ģ Within a period, elements have the same number Of Shells, But Increase in the number Valence electrons as you travel across a period. The ionization energy and the electronegativity generally increase from left to right in a row and from bottom to top in a column. As the number Of shells increase, the valence electrons are moved closer away from the nucleus. Phenomenon: Three periodic trends (atomic radius, ionization energy, and electronegativity) help chemists determine an element’s reactivity in creating compounds.Ģ Within a Group, elements have the same number of valence electrons but as you go down a group, the number of shells increase. Thus, as size (atomic radius) increases, the ionization energy should decrease.Presentation on theme: "PERIODIC TABLE TRENDS & REACTIVITY"- Presentation transcript: For larger atoms, the most loosely bound electron is located farthest from the nucleus and so is easier to remove. Because nonmetals have exactly the opposite property than that of the metals. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. The repeating properties in each row of the periodic table, as observed by Mendeleev and others, reflect the repeating electron configurations in subsequent rows. The energy required to remove the third electron is the third ionization energy, and so on.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
